

Scientific intelligence platform for AI-powered data management and workflow automation


Scientific intelligence platform for AI-powered data management and workflow automation

Objective: CGD is a rare immune system disorder characterized by recurrent pyogenic infections. Suppose we want to design a new trial to assess the effect of a new experimental product on the infection rate. (Example 1).
Year: 2019
Source: Statistics in Medicine
Link: https://onlinelibrary.wiley.com/doi/abs/10.1002/sim.8335
Clinical Area: Immunology
| Sample Size Section in Paper/Protocol: |
|
Suppose we want to design a new trial to assess the effect of a new experimental The sample sizes for these designs are as follows: |
Summary of Necessary Parameter Estimates for Sample Size Calculation:
| Parameter | Value |
| Significance Level (2-Sided) | 0.1 |
| Weibull Scale | 1.1 |
| Weibull Shape | 1.2 |
| Event Rate Ratio | 0.6 |
| Dispersion Parameter (both groups) | 0.8 |
| Exponential Dropout Rate | 0.25 |
| Power | 90 |
| Minimum Treatment Period | 1 |
| Accrual Period (Design 1;Design 2) | 0;0.5 (enrollment = 0) |
| Sample Size Allocation (balanced/unbalanced) | 0.5;0.333 (1:1;1:2) |
Step 1:
Select the MTT44 Inequality Tests for the Ratio of Two Incidence Rates using Andersen-Gill Model table from the Select Test window.
This can be done using the radio buttons below or alternatively, you can use the search bar at the end of the Select Test window and search for terms such as “MTT44” or “Andersen” and select the correct table.
After selecting the table in the window below the radio buttons, select OK to open the
table.
Step 2:
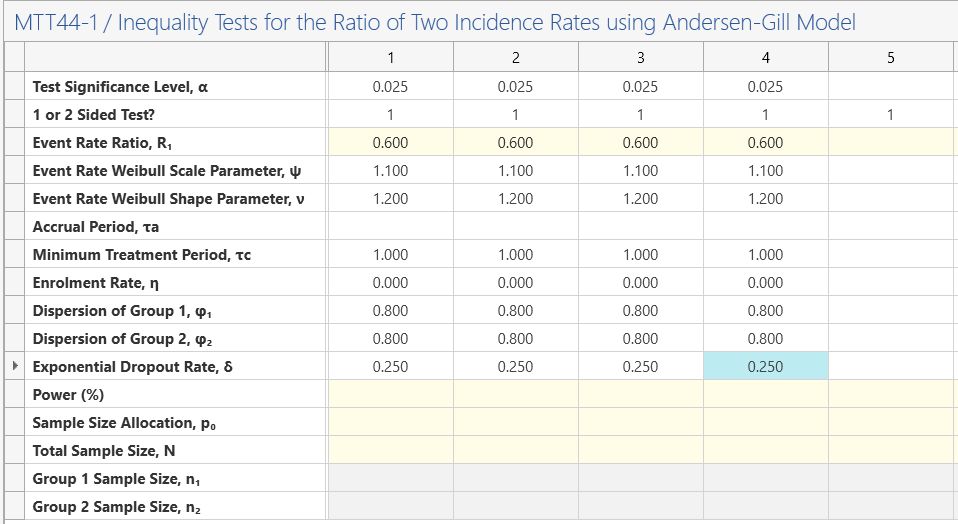
To replicate this example, the first four columns will be used for the balanced and unbalanced (1:2 controls:treatment) versions of Design 1 (no accrual period) and 2 (6 month accrual period).
The following parameters are the same in each column:
Enter the significance level of 0.025 and select a 1-sided analysis.
Enter the event rate ratio of 0.6, the Weibull Scale parameter of 1.1 and the Weibull Shape
parameter of 1.2.
As the minimum treatment period is the same for each design, enter 1 in the minimum treatment row in each column. As the enrollment rate will be ignored for the case where accrual equals zero, set this value to 0 (uniform accrual) in each column.
Enter the group 1 and 2 dispersion as 0.8 as these are assumed to be the same in each group in the study design. Enter 0.25 for the exponential dropout rate. Note this is equivalent to the δ in the design above, where 1/δ = 4 years (equivalent to 21% dropout per year).
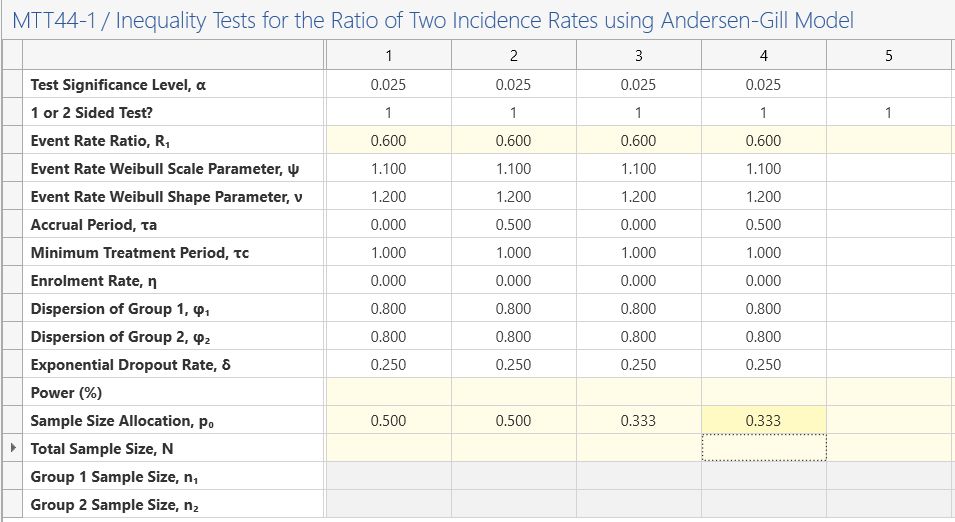
The balanced design 1, balanced design 2, unbalanced design 1 and unbalanced design 2 will be entered into columns 1-4 respectively.
In columns 1 and 3, enter an accrual period of zero. In columns 2 and 4, enter an accrual period of 0.5.
In columns 1 and 2, enter a sample size allocation of 0.5. In columns 3 and 4, enter a sample size allocation of 0.33333333.
Step 3:
Enter the power of 90% in each column and the Total Sample Size and per group sample sizes will be calculated.

| Result: |
|
The analysis results in a sample sizes of 365, 335, 390 and 360 as per Table 1 in the paper. |
We can summarize our results by using the output statement.

Copyright © Statsols. All Rights Reserved. Do Not Sell or Share My Personal Information. Privacy Policy .