

Scientific intelligence platform for AI-powered data management and workflow automation


Scientific intelligence platform for AI-powered data management and workflow automation

Objective: Teriflunomide is an oral disease-modifying therapy approved for treatment of relapsing or relapsing–remitting multiple sclerosis. We aimed to provide further evidence for the safety and efficacy of teriflunomide in patients with relapsing multiple sclerosis.
Year: 2014
Source: The Lancet Neurology
Link: http://www.sciencedirect.com/science/article/pii/S1474442213703089
Clinical Area: Neurology
| Sample Size Section in Paper/Protocol: |
|
“It was estimated that 370 patients randomly assigned to each treatment group would provide 94% power to detect a 25% relative risk reduction in annualized relapse rate, with the following assumptions: an annualized relapse rate of 0·74 for placebo; the number of relapses follows a Poisson distribution with an over- dispersion parameter of 1·3; a 1·5-year recruitment period with a linear recruitment rate (average exposure duration 1·75 years); and a 20% discontinuation rate.“ |
Summary of Necessary Parameter Estimates for Sample Size Calculation:
| Parameter | Value |
| Significance Level (2-Sided) | 0.05 |
| Expected Placebo Rate | 0.74 |
| Expected Rate Ratio | 0.75 |
| Exposure Time | 1.75 |
| Overdispersion Parameter | 1.3 |
| Covariate Distribution | Binomial |
| Proportion of Covariate | 0.5 |
| Total Sample Size (adjusted for 20% dropout) | 592 |
Note that a binomial covariate proportion of 0.5 is equivalent to equal sample size in both groups.
Step 1:
Select the “Poisson Regression” table from the Select Test Design & Goal window.
This can be done using the radio buttons or alternatively, you can use the search bar at the end of the Select Test Design & Goal window.
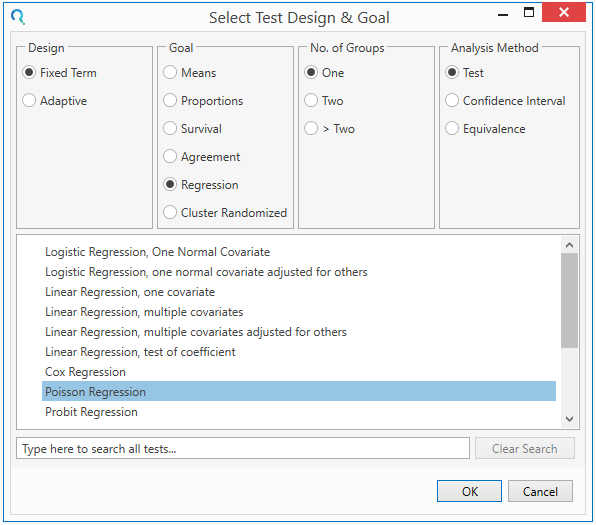
Step 2:
Enter the parameter values for the power calculation taken from the study protocol
Remember to enter “0” in the R-squared field.
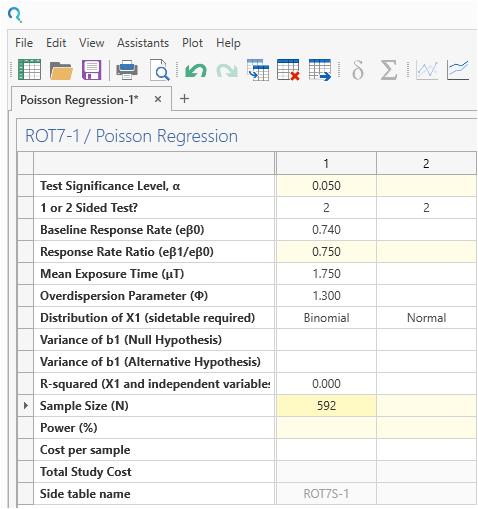
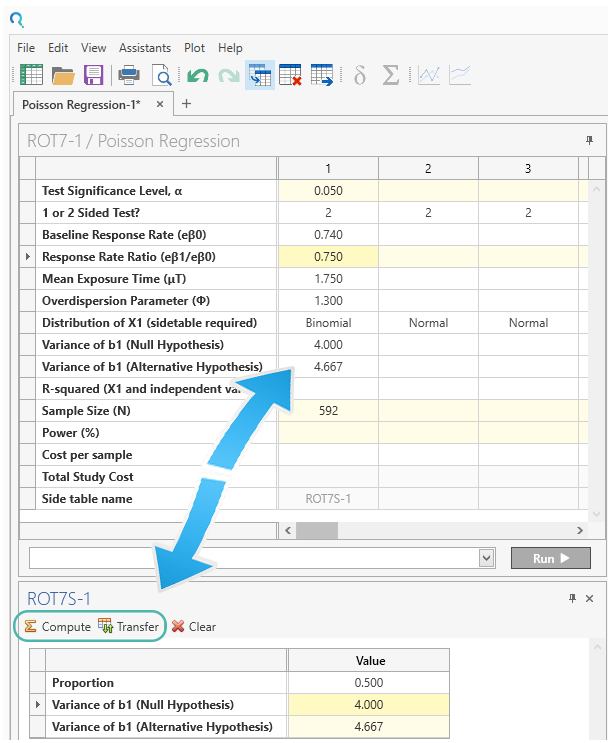
Step 3:
Enter 0.5 for “Proportion” in the binomial side table.
Click “Compute” to calculate the coefficient variances.
Then click “Transfer” to transfer them main table
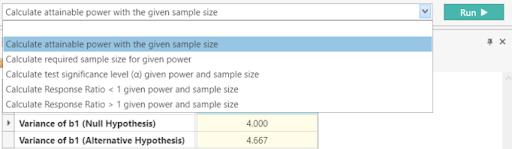
Step 4:
Beneath the table, select “Calculate attainable power with the given sample size” from the dropdown menu and click Run.
|
This gives a power of 92.2%. This is different from the stated power of 94%. However, based on the study protocol of the related TEMSO study and FDA Reviews, the power was originally calculated using a Mann-Whitney U test rather than using the Poisson Regression assumptions appropriate for the methodology used in this study. Using a sample size calculation designed for the actual statistical methodology is usually preferred. |
Copyright © Statsols. All Rights Reserved. Do Not Sell or Share My Personal Information. Privacy Policy .